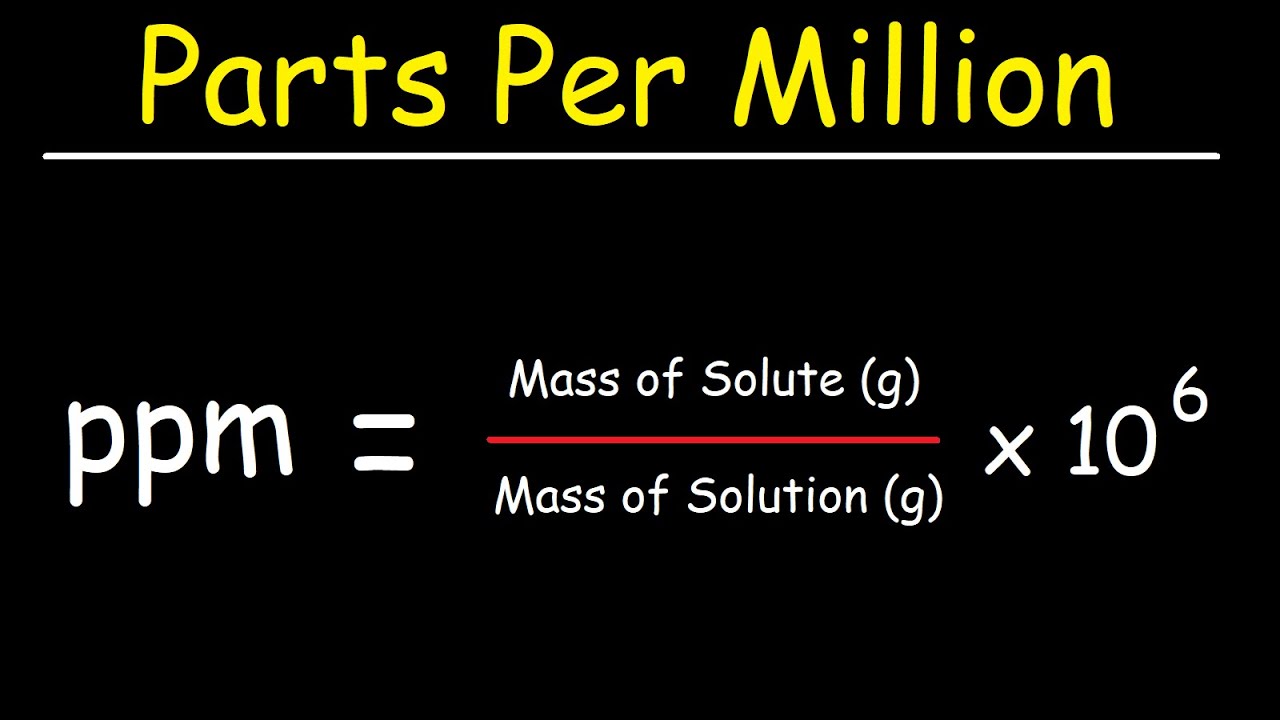
We could have used any paring of numbers that gives a mole fraction of 0.5. Using 0.5 and 1 is the simplest meaning of a mole fraction of 0.5. H 2O -> (0.5000 mol) (18.015 g/mol) = 9.0075 gĬomment: A mole fraction of 0.50 could mean 1.0 mol of one component in 2.0 total moles. Determine the mass of each mole fraction: Calculate the molality of the water:Įxample #2: Given a density of 1.769 g/mL, and a H 2SO 4 mole fraction of 0.5000, find the molality, molarity, and mass percent.ġ) We will use a mole fraction of 0.5000 to mean 0.5000 mole is present in a total of 1.0000 mole of solution. Or, the H 2O can be obtained by subtraction:ġ00.0 g / 1.836 g/mL = 54.46623 mL = 0.05446623 Lĥ) By the way, you could consider this solution to be some water (the solute) dissolved in some sulfuric acid (the solvent). Often the last mole fraction is obtained by subtraction: H 2O -> 73.44 g / 18.015 g/mol = 4.0766 molĤ) Determine the mole fraction of each component of the solution: H 2O -> 1836 g minus 1762.56 g = 73.44 gģ) Determine the moles of each component of the solution: Determine the total mass of the solution:Ģ) Determine the mass of each component of the solution: Solution assuming a certain volume of solution is present:ġ) Assume that a volume of 1.000 L of the solution is present. (Two different starting assumptions are shown.) The molar mass of water is 18.015 g/mol and the molar mass of sulfuric acid is 98.078 g/mol. Worked Examples: Converting Percentage Concentration to ppmįollow these 4 steps to convert percentage concentration to parts.ChemTeam: Calculations involving molality, molarity, density, mass percent, mole fraction Calculations involving molality, molarity, density, mass percent, mole fractionĪssume, unless otherwise told, that in all problems water is the solvent.Įxample #1: Given a density of 1.836 g/mL and a mass percent of H 2SO 4 of 96.00%, find the molarity, molality, and mole fraction. M/v (w/v) and m/m (w/w) concentrations and parts per million will be discussed in the following section (after the worked examples of percentage concentration and ppm below). V/v concentration is NOT the same as a %(v/v) concentration M/m concentration is NOT the same as a %(m/m) concentration W/v concentration is NOT the same as a %(w/v) concentration M/v concentration is NOT the same as a %(m/v) concentration Where x is the value of the percentage concentration


This is true for other similar percentage concentrations:įor mass/volume percentage concentrations (m/v% or w/v%):įor mass/mass percentage concentrations (m/m% or w/w%): We could write a mathematical expression equating parts per hundred (a percentage) and parts per million (ppm) as shown below:

How would we convert that to a concentration in parts per million (ppm) ? This means that there are 9 000 parts of NaCl in every 1 000 000 parts of the solution.īut the concentration of a solution is sometimes given as a percentage.Ī percentage concentration tells you how many parts of solute are present in 100 parts of solution.įor example, the ethanol content in wine is often given as about 12%(v/v), that is, 12% of the volume of the wine is ethanol, or, there are 12 parts of ethanol in every 100 parts of solution. Recall that, in general, concentration tells you how much solute is present in a solution.Ī concentration in parts per million (ppm) tells you how many parts of solute are present in 1 000 000 parts of solution.įor example, a saline solution is a dilute aqueous solution of sodium chloride, NaCl (aq), with a concentration of 9 000 ppm. Parts per million and Percentage Concentration Calculations
